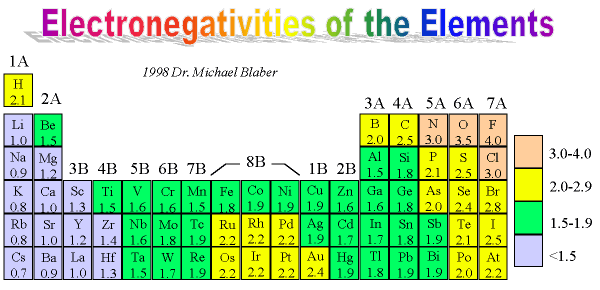
These trends explain the periodicity observed in the elemental properties of atomic radius, ionization energy, electron affinity, and electronegativity. General trend: atomic radius of elements decreases from left to right within a period, and increases from top to bottom. The radius of an atom is governed by two factors: The number of layers of electrons around the nucleus The attraction the outer electrons feel from the nucleus Compare the electronic configurations of lithium and sodium: Li: 1s 2 2s 1 Na: 1s 2 2s 2. This happens because the number of filled principal energy levels (which shield the outermost electrons from attraction to the nucleus) increases downward within each group. The chart below shows the increase in atomic radius down the group. 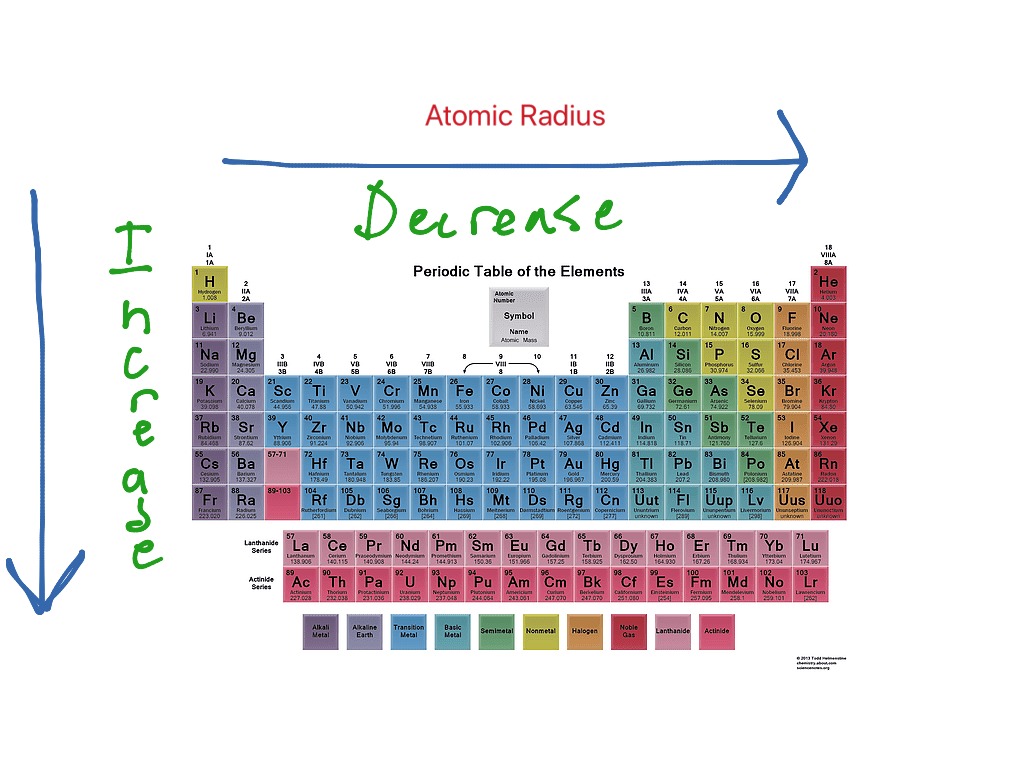
Atomic radius is one of the periodic properties of the elements. The same trend of atomic radius applies once you divide the table into metal and nonmetal sections. Ionic radius is the distance from the nucleus to the outer edge of the electron cloud of an ion. If you look at the table, you can see there is a clear trend in atomic radius. Metals Nonmetals Ionic radius increases Ionic radius increases. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Period: in general, as we go across a period from left to right, the atomic radius decreases. Second, moving down a column in the periodic table, the outermost electrons become less tightly bound to the nucleus. Atomic Radius Trend on the Periodic Table. As this happens, the electrons of the outermost shell experience increasingly strong nuclear attraction, so the electrons become closer to the nucleus and more tightly bound to it. First, electrons are added one at a time moving from left to right across a period. It assumes that you understand electronic structures for simple atoms written in s, p, d notation. In addition to this activity, there are two other important trends. Atomic radii (Clementi) Many references give table of atomic radii. This page explains the various measures of atomic radius, and then looks at the way it varies around the Periodic Table - across periods and down groups. Stable octets are seen in the inert gases, or noble gases, of Group VIII of the periodic table. Elements tend to gain or lose valence electrons to achieve stable octet formation. These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. The smallest atom on the periodic table is helium, He, and has a radius of 31 pm. 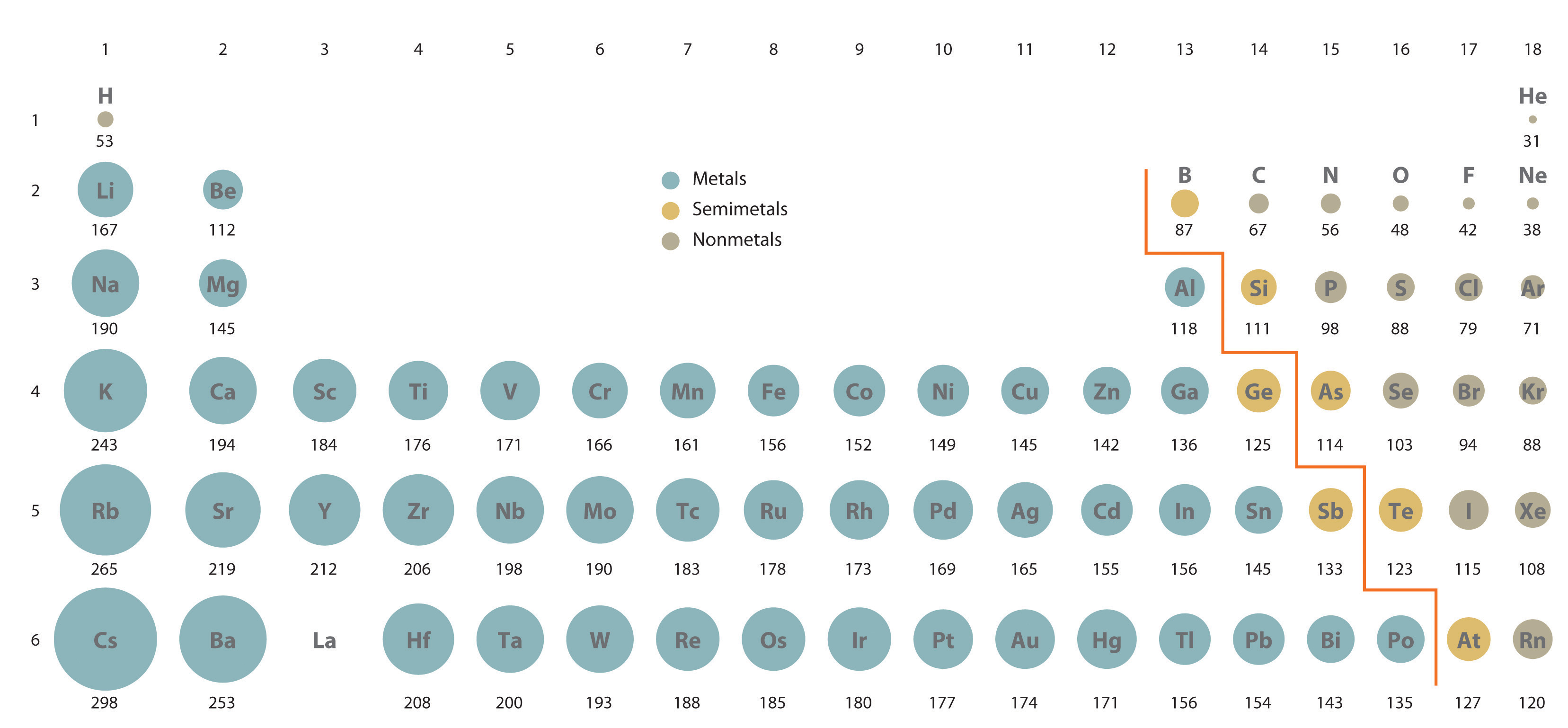
The valence electrons occupy higher levels due to the increasing quantum number (n).The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. The PDF is suitable for high-resolution color printing for desk or wall-chart display. \( \newcommand\): Periodic Table showing Atomic Radius Trendĭ own a group, atomic radius increases. The periodic table contains NIST’s latest critically evaluated data for atomic properties of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
